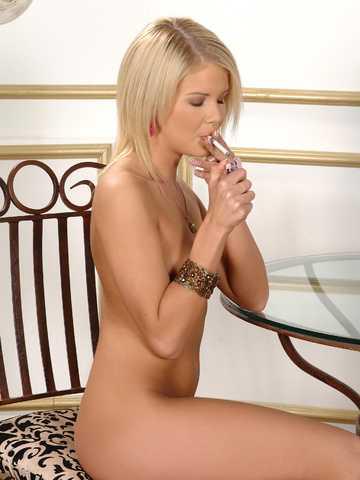Sontize

Pakolis

Felinias

Ashram

Formentera

Colore

Altum

Ekadu

Getting down to the bare minimum!

Kissers

Colore

Presenting Annis

Presenting Alina

Style

Presenting Cassandra

Maritima

Fine

Hi, I'm Cleo! I'm proud to be on

Stuffing her lunch box!

Candice

Presenting Iren

Presenting Clelia

Netes

States

Sweet Climax

Hyllie

Casandra's Mistique

Cock Craving Housewife

Cassandra's Mistique

Busty Behind Bars

Pleiades

Flexibility Is Key - Nipple Squeezing And Voluptuous Masturbation

Carnival Style Cutie

Cock Queen On Patrol - Hot Blonde Officer Action

Wide-eyed Innocence

Sultry Garden Fairy - Busty British Beauty Shows Her Big Tits

Pristine Pussy Gape - Fingering Reveals it All

Jaw-Dropping Sensation - Voluptuous Brit-Babe Masturbates For You

Her Major Mams

Sweet Climax

Heavenly Gifted: Savory Hottie Squeezes Her Big Tits

A Ravenous Snatch: Solo Pussy Play in the Day

Wet Clam Satisfaction

Presenting Cashina

Silken Skin & Giant Jugs - British Babe Masturbates On Couch

Ho Ho Ho - Blondie Fingers Herself on Christmas

Salacious Satisfaction - Hungarian Student Masturbates On Bed

Kinky Behind Bars - Cosplay Cutie Toys With A Baseball Bat

Innocent Pink Fingerings!

Her Cuteness Coaxes our Cream!

Judy's world of pink!

Boveni

Pleasing Herself and her Fans!

Shades of Blue

Pillow Fight

She'll Make You Shake

Lenfie

Gone Fishing

Blonde teen's hot striptease

A Sexy Anomaly

Praga and its perfect poonies!

Presenting Claudine

Vinota

Clearing Out The Cobwebs

White Room of Black Penetration!

Soneri

Goddess in the Garden

Fun In The Country

The Sole Attraction

Friend for Fun
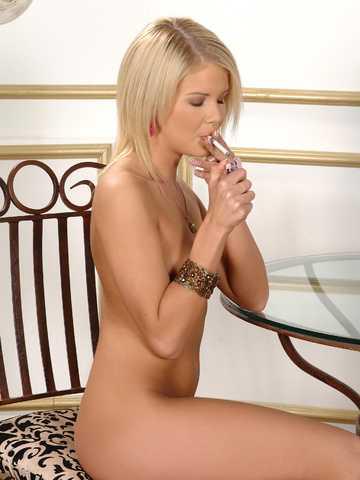
Slender blonde teen & her vibrator

Presenting Casey

Dildo Desires - A Big Glass Dick for her Pretty Slit

Nethae

Trench

Oslya

Cozy with her Toy

Fresh Face French Beauty - Absolute Stunner Makes Solo Debut

Presenting Cheyanna
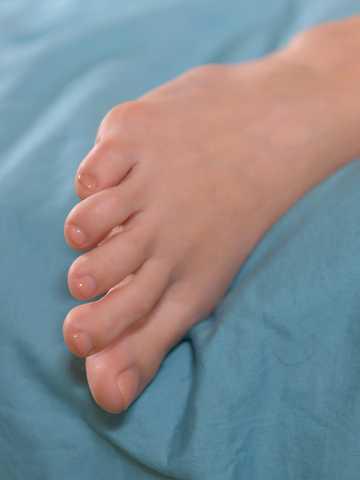
Foot Play Masturbation: Sexy Toes And Pink Pussy Fingering

Himakis

Doll to Love

Destination Load

Vaine

XXX Bubble Bath - Her Pretty Pink Gets A Cleaning

Atlantis

Summer Living

This busty blonde gets herself exci

Larewi

Beach Bunny

Pesie

Estora

Palm Fronds

Egsta

Cozy Kitchen

Cloudy Day

Naidra

Popsicle

Invigorate

Daisies

Micito

Lovely

Karas

Sexy American

Presenting Judith

Jassie: Temptation

Ladles are for labia! [Part 2]

Temptation Complete Collection

Fleur

INNOCENT AND INTRIGUING!

Plunge deep into her love! [Part 2]

Just A Simple Stunning Teen

Simple Girl

Judy Pulls Our Loads!

A butt-pluggin for her lovin!

Jassie: Just A Simple Stunning Teen Model

Juicing Up Her Jugs!

Budea

Tempting us with her backdoor!

From Pussy to Ass and back again!

Jelly doughnut anyone? [Part 2]

Jelly doughnut anyone? [Part 1]

Hot blonde Jessica Moore is just kinky enough to enjoy to present as a stream o...

Mammary marvels for ya!

The Workout Wonder

Cozy With Her Toy!

Honey Bunny - Sexy Blonde Plays With Her Pussy

Bernadett S.

Gotta Measure Up

Thelly

Untitled

Alirea

She Inspires Explosions

Curvy And Confident!

Pure Perfection

Check out newcomer's amazing butt

Choice Of Delights

Toyana

Plida

Tholes

Reale

Kimedi

Sanora

Ranice